Introduction
Blue fluorescent protein (BFP ) was first isolated from the jelly fish Aequorea victoria, this weakly fluorescent version is maximally excited at 383nm and emits at 445nm. The greatly enhanced fluorescence of Green fluorescent protein (GFP) made up of 238 amino acids and 26.9 kDa or 9nm in length is almost maximally excited at 488 nm and emits at 509nm. The green fluorescence of GFP is produced when the aequorin molecules interact with calcium ions.
Other FP molecules subsequently were developed including Yellow and Cyan FPs which are sub-optimally excited at 488nm (max at 514nm) and 405nm (max at 433nm and 452nm). The emission of YFP peaks at 530nm, whilst CFP emits maximally at 475 and 553nm respectively. Conventional flow cytometers equipped with argon and violet lasers will thus excite YFP well but sub-optimally excite CFP were a near UV laser with 440 or 473nm lines will give maximal excitation of CFP.
Other reporter molecules include the red reporters dsRED and mRFP (RED) derived from anthozoan corals and more recently Orange FP (OFP) these are sub-optimally excited by argon lasers except RFP which is like the other FPs optimally excited at or over 546nm. dsRED and mRFP emit at around 590nm whilst OFP emission peak is at 568nm.
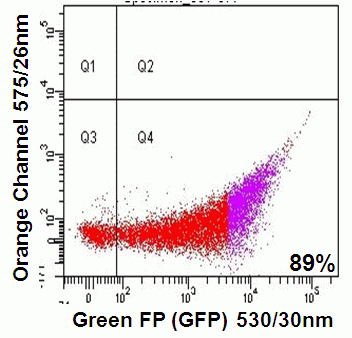
The development of 'Fruit' named reporter molecules by Tsein has again expanded the range of fluorescent gene reporters. These are divided into orange and red probes based on their maximum excitation. Most of the 'Fruit' reporters have a maximum and sub-optimal excitation peak ranging from 540-590nm and 510-560 respectively. The emission peaks are normally close to the excitation peak. For example the orange based dTomato excites maximally at 554nm (sub at 518nm) and emits at 581nm; mBanana ex 556 em 610nm; mCherry ex 587nm em 610nm. The Millipore Muse 532nm green laser line can effectively excite mCherry with emission collected in the first fluorescence parameter at 575nm, see figure.
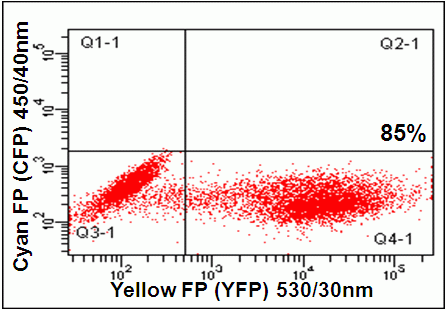
GFP can be analysed with YFP on flow cytometers with appropriate filter and mirror changes. The GFP detector on a BD instrument should have a 502LP mirror with a 510/10nm BP filter. While the PE 585/42nm channel which normally has a 556LP mirror should be changed to detect YFP by installing a 525LP mirror and a 540/40nm BP filter.
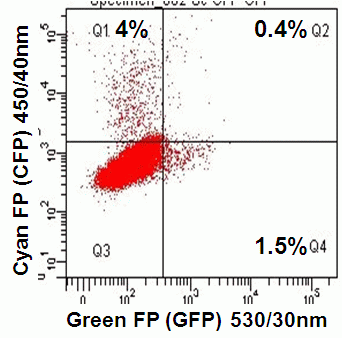
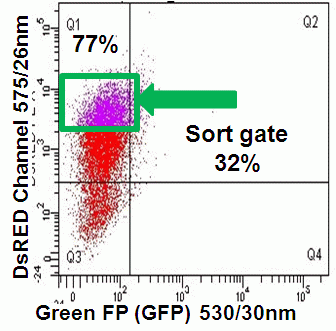
Likewise GFP with peak emission at 509nm can be run in conjunction with FITC or it's analogues such as CDFA the hydrogen peroxide detecting dye. The FITC 530/30nm channel is switched with a 510/10nm BP filter to detect GFP. While the PE 585/42nm channel should be changed to 530/30nm BP filter fitted with a 525LP mirror, see figure.
For an exhaustive list of available FP proteins see the attached pdf from i-cyt.com