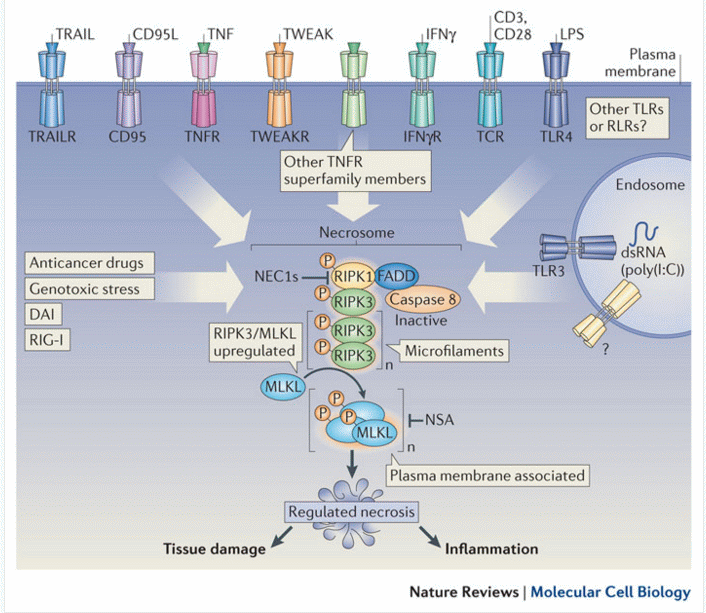
Necroptosis is a programmed cell death process occurring in vertebrate cells in response to infection by pathogens which inhibit the classic programmed cell death process apoptosis. Pathogens block apoptosis by their anti-capsase activity resulting in signalling to the necrosome resulting rapid cell death by necroptosis, thus minimizing the spread of the pathogen. other forms of necrotic cell death such as oncosis by a nonphysiological event resulting in cell swelling as opposed to apoptosis where there is cell shrinkage or necroptosis were there is controlled leakage of the cell contents through the plasma membrane.
First, extrinsic stimulus through the TNF receptor by TNFα signals the recruitment of the TNF receptor-associated death domain (TRADD) which in turn recruits RIPK1. In the absence of active Caspase 8, RIPK1 and RIPK3 auto- and transphosphorylate each other, leading to the formation of a microfilament-like complex called the necrosome. he necrosome then activates the pro-necroptotic protein MLKL via phosphorylation. MLKL actuates the necrosis phenotype by inserting into the bilipid membranes of organelles and plasma membrane leading to expulsion of cellular contents into the extracellular space, see figure. The inflammatory rupturing of the cell releases Damage Associated Molecular Patterns (DAMPs) into the extracellular space. Many of these DAMPs remain unidentified, however, the “find me” and “eat me” DAMP signals are known to recruit immune cells to the damaged/infected tissue. Necrotic cells are cleared from the immune system by a mechanism called pinocytosis, or cellular drinking, which is mediated by macropinosomes, a subcellular component of macrophages. This process is in contrast to removal of apoptotic cells by the immune system in which cells are removed via phagocytosis, or cellular eating.
The 'Intrinsic pathway' can be drug induced, UV-B irradiation, g-irradiation, and reactive oxygen and is mediated via mitochondrial responses to such cellular insults, see model below:-
Classic apoptotic cells can be recognized by a characteristic pattern of morphological, biochemical and molecular changes, which may be broadly and chronologically defined as:
Morphological Changes- Cell shrinkage
- Cell shape change
- Condensation of cytoplasm
- Nuclear envelope changes
- Nuclear fragmentation
- Loss of cell surface structures
- Apoptotic bodies
- Cell detachment
- Phagocytosis of remains
- Free calcium ion rise
- bcl2/BAX interaction
- Cell dehydration
- Loss of mitochondrial membrane potential
- Proteolysis
- Phosphatidylserine externalisation
- Formation of Mitochondrial Permeability Transition Pore
- Lamin B proteolysis
- DNA denaturation
- 50-300kb cleavage
- Intranucleosomal cleavage
- Protein cross-linking
Many of these changes may be measured by flow cytometry and some are described below.
Publications.
A Vossenkamper, G Warnes. Thapsigargin induced apoptosis occurs with a delayed DNA damage response, Br J Cancer Res 2(4):314-319, 2019. (in press ).
A Vossenkamper, G Warnes. Flow cytometry reveals the nature of oncotic cells, Int. J. Mol. Sci. 20(18), 4379; 2019.
D Bergamaschi, A Vossenkaemper, WYJ Lee, P Wang, E Bochukova, G Warnes. Simultaneous polchromatic flow cytometric detection of multiple forms of regulated cell death. Apoptosis, 2019. (DOI: 10.1007/s10495-019-01528-w)
A Popat, AA Patel, G Warnes. A flow cytometric study of ER Stress and autophagy, Cytometry A, DOI: 10.1002/cyto.a.23665, 2018.
A Vossenkamper, G Warnes. A flow cytometric immunophenotyping approach to the detection of regulated cell death processes. Journal of Immunological Sciences, 2 (5) 2018.
HL Lee, R Pike, MHA Chong, A Vossenkamper, G Warnes. Simultaneous flow cytometric immunophenotyping of necroptosis, apoptosis and RIP1 dependent apoptosis Methods, 1, 134-135, 56-66, 2018.
Luo J, Lee S, Wu D, Yeh J, Ellamushi H, Wheeler AP, Warnes G, Zhang Y, Bo X. P2X7 purinoceptors contribute to the death of Schwann cells transplanted into the spinal cord. Cell Death Dis. Oct 3;4:e829. doi: 10.1038/cddis.2013.343, Oct 2013.
TS MacFie, R Poulsom, A Parker, G Warnes, TBoitsova, A Nijhuis, N Suraweera, APoehlmann, JSzary, R Feakins, R Jeffery, RW Harper, AM Jubb, JO Lindsay, A Silver. DUOX2 and DUOXA2 form the predominant enzyme system capable of producing the reactive oxygen species H2O2 in active ulcerative colitis and are modulated by 5-aminosalicylic acid. Inflammatory Bowel Disease, Jan 2014.
SN Jayasinghe, G Warnes, CJ Scotton. Bio-electrosprayed living microenvironments implanted into mouse models. Macromolecular Bioscience 11(10), 1364-69, 2011.
SM Janes, TA Ofstad, DH Campbell, A Eddaoudi, G Warnes, D Davies, F Watt. PI3-kinase dependent activation of apoptotic machinery occurs on commitment of epidermal keratinocytes to terminal differentiation. Cell Res 19(3): 328-339, 2009.