A dot plot of forward light scatter versus 90o side scatter is a measure of cell size and cell granularity respectively, the latter being dependent upon the presence of intracellular structures that change the refractive index of light. Cell condensation during early apoptosis is seen as a decrease in forward light scatter only. As apoptosis proceeds, both forward and 90o side scatter diminish. In necrotic death, cells swell initially, and then cellular contents are rapidly released. This is seen on the flow cytometer as an initial increase in forward light scatter followed by a rapid decrease in both forward and 90o side scatter. Changes in cell size can be used in conjunction with surface phenotypic markers to identify the dying population.
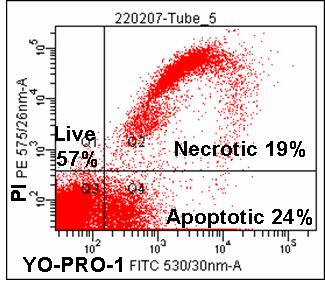
Light scatter and various dyes may also be used. These include DAPI (350-60/405nm excitation; violet fluorescence emission); Sytox Green (488nm excitation; green fluorescence emission), propidium Iodide (488nm excitation; orange/red fluorescence emission), 7-Aminoactinomycin-D (7-AAD) (488nm excitation; red fluorescence emission); SYTOX ADDvanced (488nm excitation; red fluorescence); DRAQ7 (488 and 633nm excitation; red fluorescence emission) and TO-PRO-3 (633nm excitation; red fluorescence emission). For example, membrane rupture seen in necrosis and the very late stages of apoptosis, permeabilises cells to propidium iodide.
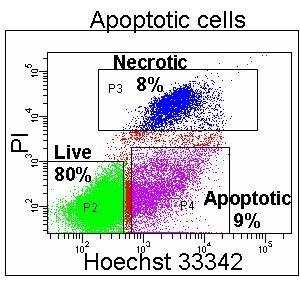
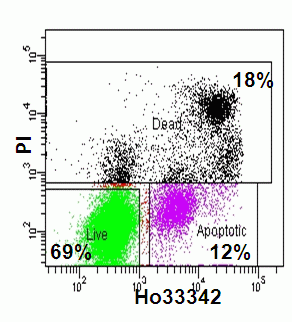
However, certain changes in energy dependent ionic membrane transport during apoptosis allows a degree of fluorochrome up-take for a dye such as 7-AAD. This permits a discrimination of viable cells which exclude the dye totally, from apoptotic cells that fluoresce dimly and necrotic and late apoptotic cells that fluoresce brightly. Likewise Hoechst 33342 or YO-PRO-1 can be used in conjunction with PI if a short incubation time is applied to the cells of interest the DNA dyes preferentially load into the apoptotic whose plasma membranes are becoming compromised compared to healthy unaffected cells