CFP-YFP FRET Analysis
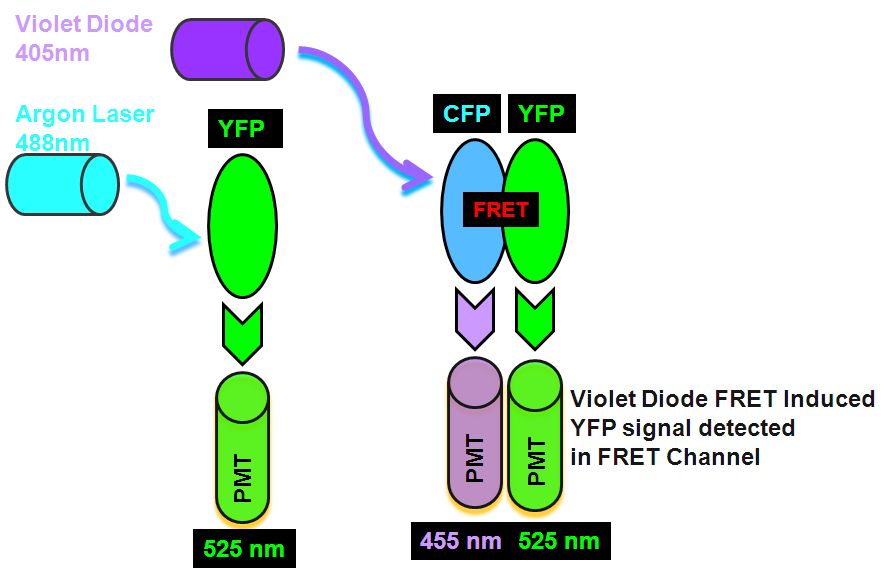
CFP-YFP FRET between two proteins is widely used to investigate cell process, even though this technique has difficulties as the degree of FRET occurring between two proteins is dependent upon spatial orientation and proximity. Recently the development of genetically encoded dual fluorophores such as CFP and YFP, thus serving as built in Donor and Acceptor for FRET detection when the protein undergoes phosphorylation of calcium binding, see figure.
This type of FRET sorting of live cells can be done on a instrument with an violet diode and argon lasers, these two lasers excite CFP and YFP separately. Firstly the argon 488 nm laser excites the YFP which emits light in the argon laser 530 nm PMT. Next, violet diode excitation (405nm) of the Donor, CYP transfers energy non-radiatively to the Acceptor, YFP which then emits FRET derived energy at 530 nm in the violet diode 530nm PMT, see figure for pictorial explanation in Theory of FRET detection. The median fluorescence's of non-FRET control and FRET sample at channel 2,137 and 31,700, gives a FRET Efficiency Ratio of 13.8 according to the FRET Efficiency Ratio Formula.
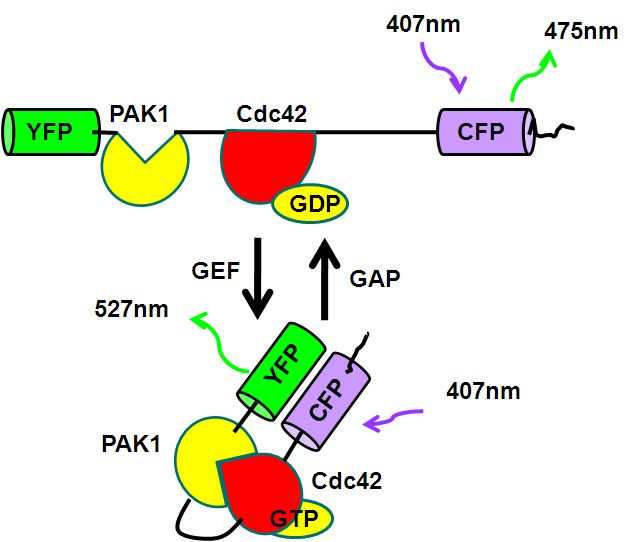
The protein Cdc42 is member of the Rho family G proteins and triggers filopodium formation during cell movement and is regulated by guanine nucleotide exchange factor (GEF) and GTPase-activating protein (GAP). GEF promotes the exchange of GDP to GTP resulting Cdc42 binding to PAK1 bringing YFP closer to CFP and hence a greater FRET signal which can be detected and quantitated flow cytometrically, see figure.
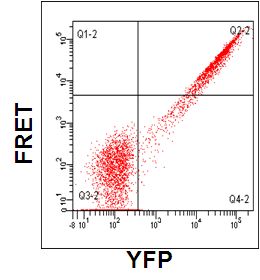
Single CFP and YFP colour mutants are used to set compensations for YFP and CFP bleed through to 530nm Argon (YFP) and violet channels (FRET) and 450nm violet channel (CFP), see figure. Cells expressing Cdc42-CFP-YFP show a high degree of FRET, with a ratio of 13.8, see figure.