CFP-YFP FRET Sorting
Cell cultures of cells transfected for two proteins labelled with CFP and YFP often have low levels of expression and only a small population of cells will be capable of FRET given the level of expression of the two proteins of interest, see figure.
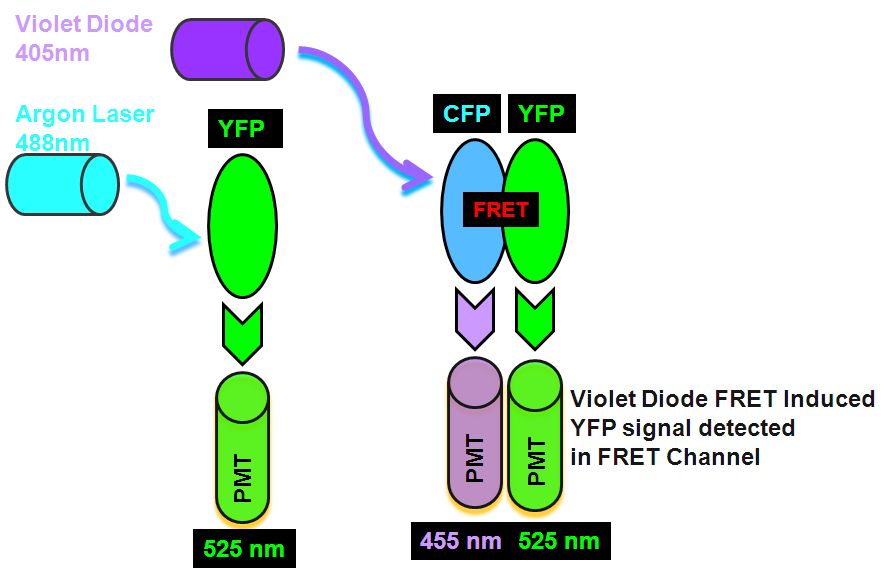
The sorting of such live cells demonstrating FRET can be done to improve the level of FRET detectable in cultures before imaging of FRET by microscopy. An Aria I cell sorter instrument fitted with a violet diode and argon laser, firstly excites the YFP with 488 nm laser which emits in the argon laser 530 nm PMT. Next, violet diode excitation (405nm) of the Donor, CYP transfers energy non-radiatively to the Acceptor, YFP which then emits FRET derived energy at 530 nm in the violet diode 530nm PMT, see figure for pictorial explanation in Theory of FRET detection.
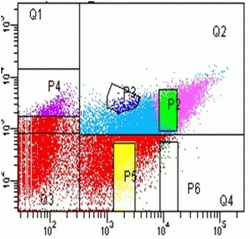
Single CFP and YFP colour mutants are used to set compensations for YFP and CFP bleed through to 530nm Argon (YFP) and violet channels (FRET) and 450nm violet channel (CFP), see figure.
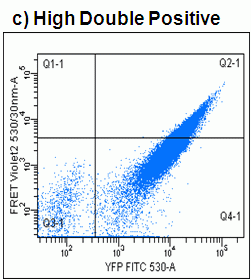
After sorting low and high double positives for CFP and YFP the FRET signal achieved was 26% in the high expressors compared to 10% displayed by the original culture of cells, see figure.
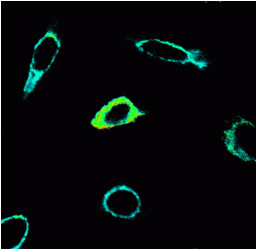
The high double positive culture of cells was then imaged on a Zeiss 510 confocal microscope. CFP and YFP was excited by the 405 and 514nm laser lines. Images of FRET were produced by the Sensitization method and FRET images calculated via the Zeiss FRET software, see figure.