Organelle function - mitochondrial membrane potential (Δψm) changes
The Δψm is due to a differential distribution of proteins on either side of the impermeable inner mitochondrial membrane. In oncosis the loss of Δψm is dramatic compared to the loss observed during apoptosis. This observation is reflected in the energy demands of oncosis and apoptosis upon the cell, oncosis being ATP independent and does not require mitochondrial function to proceed. Whilst apoptosis is ATP dependent and requires a degree of mitochondrial function during the apoptotic process.
The fall in mitochondrial inner membrane potential can be measured flow cytometrically. Normal mitochondrial function has a requirement for a negative charge on the inner side of the mitochondrial inner membrane. This is maintained by the asymmetrical distribution of H+ ions across the membrane giving rise to both a potential difference and a chemical pH gradient. Cationic lipophilic dyes such as CMXRos (chloromethyl-X-rosamine), TMRE (tetramethylrhodamine), JC-1, DiOC6(3), DiIC1(5) and rhodamine 123 localize across the inner mitochondrial membrane a process which is dependent upon H+ distribution.
Loss of mitochondrial membrane potential is detected in cells by measuring a loss of dye from the inner mitochondrial matrix or a change in fluorescence of the dye as the transmembrane potential drops. Oncosis can be induced by chemical, detergent, heat shock and high dose drug treatments. Below are some examples of these treatments and their affect upon mitochondrial function over a 4 hour period.
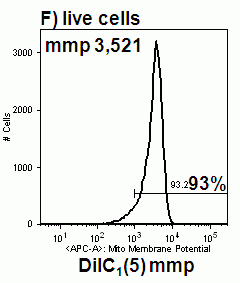
Chemical induction of oncosis by 1% sodium azide causes Jurkat cells to lose mitochondrial function rapidly. Labelling such cells with annexin V-FITC, DAPI and DiIC1(5) shows that oncotic cells lose mitochondrial function, see figure.
Detergent induction of oncosis by 0.01% Triton X-100 causes Jurkat cells to lose mitochondrial function rapidly. Labelling such cells with annexin V-FITC, DAPI and DiIC1(5) shows that oncotic cells lose mitochondrial function, see figure.
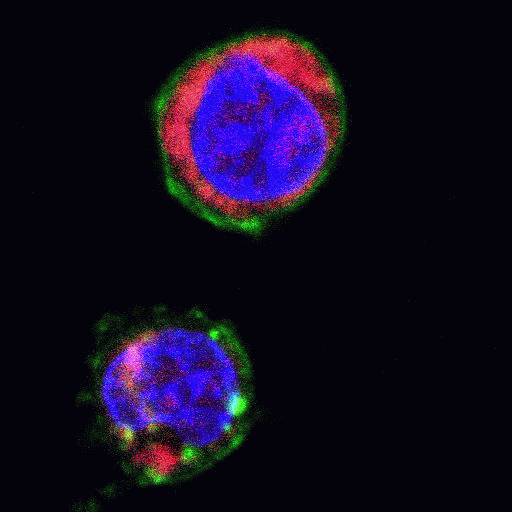
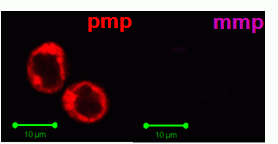
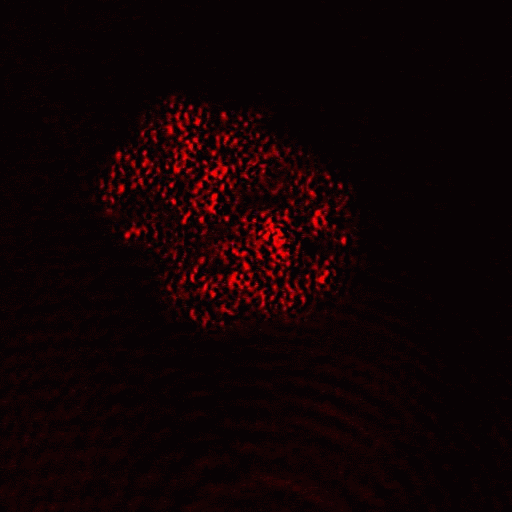
Heat shock induction of oncosis causes Jurkat cells to lose mitochondrial function more rapidly as the temperature is increased from 42C to 56C. Labelling such cells with annexin V-FITC, DAPI and DiIC1(5) shows that oncotic cells lose mitochondrial function, see figure for heat shock at 42C and 56C respectively. Dead cells after heat shock at 56C treatment appear to have a normal mitochondrial function, however imaging of such cells show a very diffused staining pattern of the carbocyanine dye indicating that mitochondria have been disrupted, see figure for comparison. A higher magnification image of DiIC1(5) stained mitochondria in a live cells is shown in the figure.
Real-time membrane potential changes for the kinetic analysis of oncosis
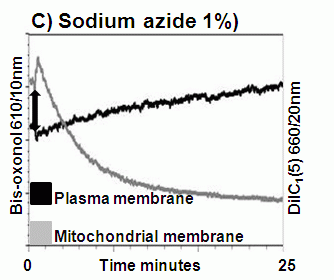
Apoptosis is an ATP dependent process whilst oncosis is not, this fact can be used to differentiate between the two processes in a real-time manner by flow cytometric analysis of mitochondrial membrane potential by use of carbocyanine dye DiIC1(5). The different effects induced by the oncotic and apoptotic processes upon cell membranes of the cell this is especially true of the plasma membrane. The potential of the plasma membrane can be measured in a real-time manner by use of the Oxonol dye, DiCAB4 or bis-oxonol, see figure.
Oncotic agents, such as heat shock, sodium azide and Triton X-100 induce a rapid detectable reduction in mitochondrial function and plasma membrane depolarization; as shown by a rapid fall and increase in DiIC1(5) and bis-oxonol intensity respectively. Whereas induction of apoptosis results only in small insignificant changes in membrane potentials. These changes in membrane potentials during oncosis can be observed by confocal microscopy, see figure.
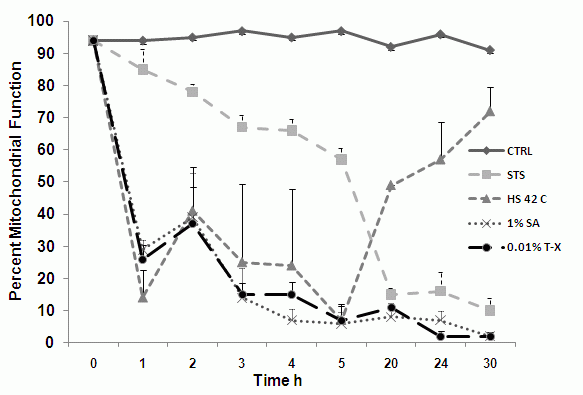
Time-dependent analysis of mitochondrial function during onosis
Mitochondrial function can be monitored at hourly times showing that mitochondrial function is lost in a gradual manner during apoptosis and is rapidly lost during oncosis induced by heat shock (42C), 1% sodium azide and 0.01% Triton X-100 treatments, see figure.